 Since the quantity of reactants on the left-hand side (LHS) mostly differs from the quantity of the product/s on the right-hand side (RHS), a skeleton equations is said to be an unbalanced equation. Thus, the skeleton equation shows only the elements which are involved in a chemical reaction, but does not express the quantities in which they react or are produced. One chlorine atom isn’t accounted for by the skeleton equation. However, if you carefully observe the above example equation, you will notice that, even though there are two chlorine atoms on the reaction side, there is but a single one on the product side. The skeleton equation thus provides us with a better means of expressing a chemical reaction, as compared to a word equation. 
The following is an equation for the reaction that produces salt: This increases the utility of the skeleton equation. The symbol (s) is used to represent a solid, (l) a liquid, (aq) an aqueous, and (g) a gas. Thus, a chemical reaction can be expressed as:įurther, in skeleton equations, the physical states of the reactants and products are mentioned as subscripts on the right-hand side next to each element. The products are the molecules that are obtained as a result of a chemical reaction. The reactants are the original elements (atoms and molecules), which, under suitable conditions, react chemically with each other. The most basic elements of any chemical reaction are the reactants and the products. Thus, for example, while freezing water to ice is merely a physical process, the chemical combination of sodium and chlorine gas which results in the formation of sodium chloride (salt) is a chemical reaction. In chemical reactions, atomic/molecular bonds are formed, broken or rearranged, so that a chemically different product is formed. It is important to note that, chemical reactions are different from processes like melting or freezing, which are essentially physical processes wherein only the state of the matter changes. In chemistry, a chemical reaction is said to have taken place when two or more elements react with each other to produce a new element/s. For this purpose, scientists around the world make use of chemical equations. 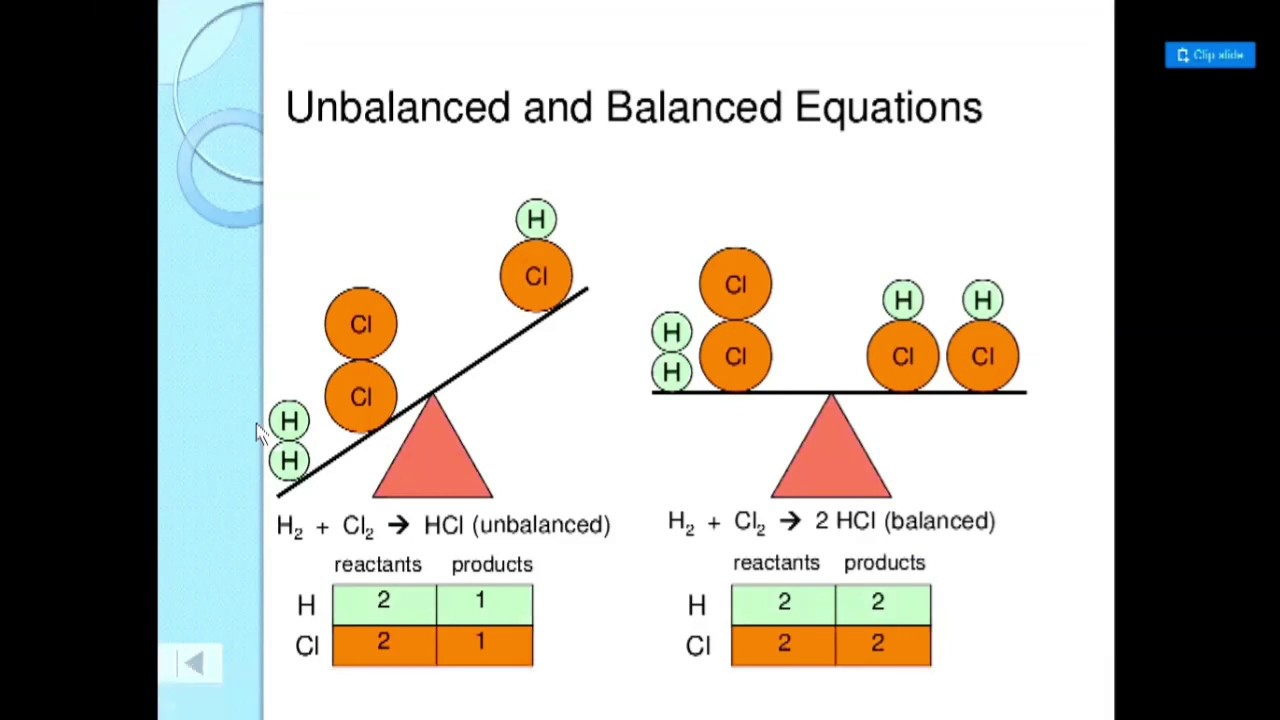
Needless to say, therefore, it is of utmost importance that we study how these important chemical reactions take place.īut to be able to study the literally thousands of possible chemical reactions, there needs to be a proper way of representing them in writing, which is in accordance to the laws of nature, and which is mathematically correct. These basic forms of matter react with each other chemically to form all the complex living and non-living things that we see around us. We ourselves and indeed the whole world are made of the 118 identified elements in the periodic table. The rest of the elements can be represented in the mono-atomic form. Therefore, one must take care to use proper formula for these elements while substituting them in skeleton equations. 
Other diatomic elements are H2, F2, Cl2, Br2, I2, O2, and N2―all of which are gases. Oxygen is a diatomic element which is always found as O2 in nature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
